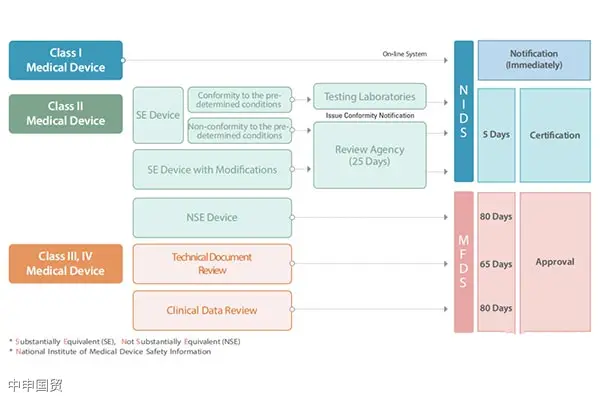
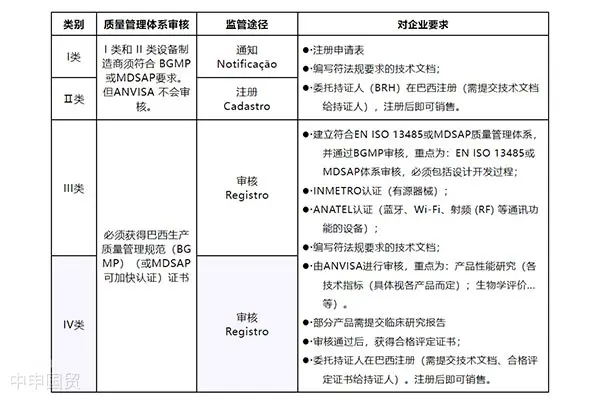
Certification requirements and procedures for exporting medical devices to Canada
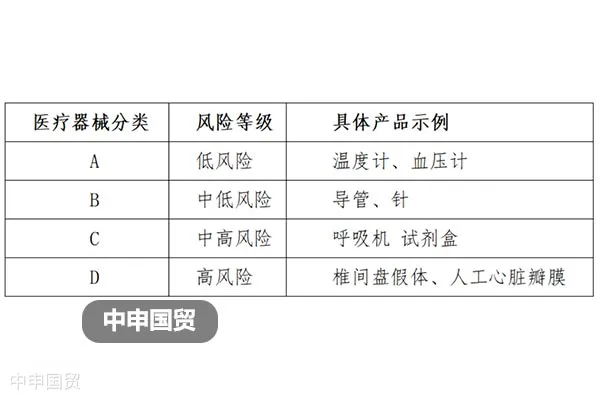
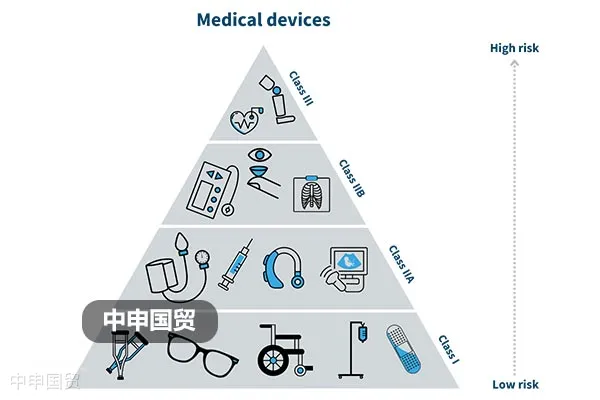
In Canada, the market access of medical devices is strictly regulated. In addition to meeting the requirements of the Food and Drugs Act and the Medical Devices Regulations, it also involves the classification of medical devices, license requirements, labeling regulations, and restrictions on illegal marketing.










 PSB Record: Shanghai No.31011502009912
PSB Record: Shanghai No.31011502009912