- Shanghai Zhongshen International Trade Co., Ltd. - Two decades of trade agency expertise.
- Service Hotline: 139 1787 2118
Medical EquipmentExporting to Malaysia requires meeting some specific certifications and requirements. The following are the certifications and precautions for exporting medical devices to Malaysia:
I. Medical Device Legislation and Competent Authorities
Malaysias Medical Device Act is the Medical Device Act (2012), and its basic framework is relatively similar to the US medical device regulations. The management departments of medical devices include the Medical Device Authority (MDA) of the Ministry of Health and the Atomic Energy Licensing Board of the Ministry of Science and Technology. The Medical Device Authority of the Ministry of Health is responsible for managing all medical devices except radioactive medical devices and second - hand medical devices, while the Atomic Energy Licensing Board is responsible for managing radioactive medical devices and second - hand medical devices.
II. Market Access Process for Importing Medical Devices in Malaysia
(1) Definition and Classification of Medical Devices in Malaysia
Medical devices are defined as any instrument, apparatus, implement, machine, implant, in - vitro reagent or calibrator, software, material, etc., used alone or in combination for the following purposes. They can be used for diagnosis, prevention, monitoring, treatment, alleviation of diseases and injuries, research on anatomical or physiological processes, support or maintenance of life, disinfection of devices, and providing information for medical or diagnostic purposes through in - vitro examination of specimens taken from the human body. However, if these functions are exerted through processes such as drugs or immunization, it cannot be defined as a medical device. According to Malaysias medical device regulations, medical devices are classified into four categories: Class A, Class B, Class C, and Class D from low to high risk. Among them, Class A medical devices have the lowest risk, Class B and Class C are in the middle, and Class D has the highest risk.
Class A products: Low - risk, such as cell counters.
Class B products: Medium - low risk, such as urine analyzers.
Class C products: Medium - high risk, such as electrocardiographs.
Class D products: High - risk, such as hemodialysis machines.
(2) Classification and Registration of In - vitro Diagnostic Reagents
In - vitro diagnostic reagents (IVD) refer to devices used alone or in combination mainly or only to provide diagnostic and monitoring information for the human body, including reagents, calibrators, sample containers, etc. According to the Medical Device Act, IVD products are classified into four categories: A, B, C, and D according to the level of impact on personal and public health risks. The risk levels and product examples of each category are as follows:

For the registration of IVD, it is necessary to follow the special applicable guidelines in the Medical Device Act and register according to the corresponding steps.

III. Registration Process for Importing Medical Devices in Malaysia
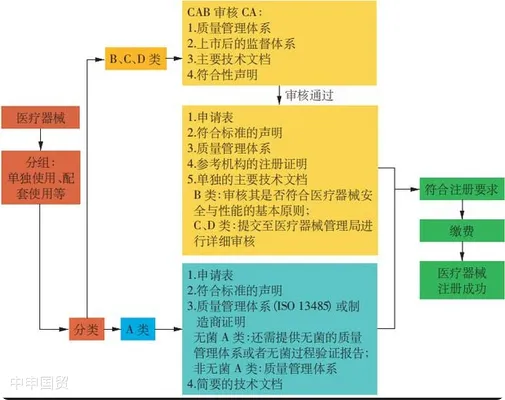
(1) Class A Products
For Class A products, the local authorized representative of medical devices (AR) can apply for registration to the Medical Device Authority of the Ministry of Health without the approval of a conformity assessment body (CAB) authorized by MDA. The applicant needs to submit documents such as the manufacturers ISO 13485 certificate, test report, and label.
(2) Class B, C, D Products
For Class B, C, D products, the local authorized representative of medical devices needs to submit a technical report, and the technical documents will be reviewed by a conformity assessment body. Medical devices that have been approved and sold in reference countries (such as Australia, Canada, the EU, Japan, the US) can be reviewed through a simplified procedure. During the review process, documents such as ISO certificates and CE certificates need to be submitted to the conformity assessment body. After passing the review, the conformity assessment body will issue a certificate. The final device registration documents, including the Common Submission Dossier Template (CSDT), the conformity assessment body certificate, and the application documents, will be submitted electronically online to the Medical Device Authority of the Ministry of Health for review and final approval. The certificates of the conformity assessment body and the device registration certificate should be renewed every 5 years.
IV. Precautions
In the process of exporting medical devices to Malaysia, the following points need to be noted:
(1) Currently, the Free Sales Certificate (FSC) is no longer required for registration in Malaysia.
(2) The registration application must be submitted at the designated local representative office in Malaysia.
(3) Each product can only have one product license holder (i.e., the licensee).
(4) The product license can be transferred to other holders.
(5) All manufacturers need to obtain ISO 13485 certification as a necessary condition for applying for registration.
In summary, exporting medical devices to Malaysia requires meeting the registration requirements stipulated by the Medical Device Administration Law. The specific process includes classification determination, technical document review and submission, conformity assessment body certification, and final online registration approval. Before applying for registration, relevant regulations and requirements should be carefully understood to ensure compliance with Malaysias medical device export standards.
Related Recommendations
Category case
Contact Us
Email: service@sh-zhongshen.com
Related Recommendations
Contact via WeChat

? 2025. All Rights Reserved. 滬ICP備2023007705號-2  PSB Record: Shanghai No.31011502009912
PSB Record: Shanghai No.31011502009912