- Shanghai Zhongshen International Trade Co., Ltd. - Two decades of trade agency expertise.
- Service Hotline: 139 1787 2118
This article provides a detailed analysis of the certifications and procedures required for exportingMedical Equipmentto India, including the regulatory authorities for medical devices in India, the classification of medical devices, the application for a manufacturing license for medical devices exported to India, the regulations on medical device labels, and the specific steps of the process for exporting medical devices to India.
I. Regulatory Authorities for Medical Devices in India
The regulation of medical devices in India is the responsibility of the Central Drug Standards Control Organization (CDSCO) under the Ministry of Health & FW. An important department within this agency is The Drug Controller General of India (DCGI), which is responsible for formulating the national medical device policy.
II. Classification of Medical Devices
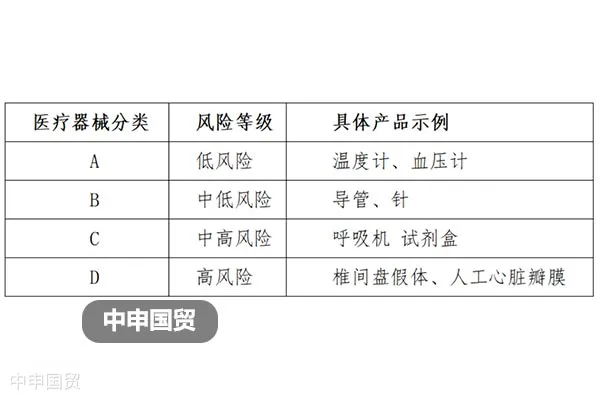
In 2017, India announced the Medical Device Rules 2017. Referring to The Global Harmonization Task Force (GHTF), medical devices are classified into four categories from A to D, corresponding to low to high risk levels.
III. Application for a Manufacturing License for Medical Devices Exported to India
Medical devices exported to India must be applied for an import license by the local authorized agent in India to CDSCO. The application process mainly includes the following two steps:
Confirmation of the compliance of the manufacturers Quality Management System (QMS): First, CDSCO will review the manufacturers QMS submitted by the authorized agent, which must comply with the Indian medical device quality management standard ICMED13485 (modified from ISO13485, and Chinas standard YY/T0287 is equivalent to ISO13485). This review includes the manufacturers QMS technical documents, test reports, the latest on-site inspection report, etc. If CDSCO has doubts about the compliance of the manufacturers QMS, it may require an assessment, product testing or an on-site inspection of the manufacturer (the cost shall be borne by the agent).
Review of the safety and effectiveness of medical devices: If the medical devices exported to India are classified as Class A or Class B, the authorized agent should submit a Free Sale Certificate from China to CDSCO, or data from clinical tests implemented in China (or other data available to prove the safety and effectiveness of the product). If the medical devices exported to India are classified as Class C or Class D, the authorized agent must conduct clinical tests in India. Exception: If the medical device (Class A to Class D) has been issued a Free Sale Certificate by the regulatory authorities of the EU, the US, Canada, Japan or Australia, no further clinical tests are required.
The entire license application process requires meticulous and precise work, including preparing and submitting a full set of documents, tracking and answering possible questions, and handling possible on-site inspections and evaluations. During the entire process, it is very important to maintain good communication and interaction with CDSCO to ensure the smooth progress of the application process. At the same time, having an in - depth understanding of Indian medical device regulations is also a key factor in ensuring the success of the application.
IV. Medical Device Labels
The Medical Device Rules 2017 in India have strict regulations on medical device labels. Articles 44 to 48 stipulate the essential content of the label:
(1) Name of the medical device
(2) User identification device and details necessary for its use
(3) Name of the manufacturer and the address of the manufacturing site where the device is manufactured
(4) An accurate statement of the net quantity, which shall be expressed in metric units in terms of weight, dimensions, volume, number of units (as the case may be) and the number of devices contained in the package
(5) Manufacturing date and expiration date (or the label shall indicate the shelf life of the product)
(6) Provide instructions, if necessary, to indicate that the device contains medicinal or biological substances.
(7) Provide a distinct batch number, starting with the term Lot No., or Lot, or Batch No., or B.No..
(8) Indicate, if necessary, any special storage or handling conditions applicable to the device.
(9) Specify whether the device is provided as a sterile product, its sterile status, and the sterilization method.
(10) Give warnings or precautions (if deemed relevant) to draw the attention of the users of the medical device.
(11) If the device is for single - use only, the device should be appropriately labeled.
(12) If the medical device is intended to be distributed as a free sample to medical professionals, overprint the words Physician’s Sample—Not to be sold on the label of the device.
(13) Except for imported devices, add the manufacturing license number before Manufacturing Licence Number or Mfg. Lic. No. or M.L.
(14) If it is an imported device, provide (if not already printed) such details on the label by pasting, including: import license number, the name and address of the importer, the address of the actual production site, and the manufacturing date.
(15) If small medical devices, due to their size, cannot clearly print all the information, at least the information required for product identification and safety should be included.
V. Process Steps for Exporting Medical Devices to India
The general steps for exporting medical devices to India include:
(1)Understand Indias regulatory requirements:Research and understand Indias regulations and requirements for medical device exports, including registration, licenses, labeling requirements, quality control standards, etc.
(2)Registration and certification:Register and obtain certification in India to prove that the product meets local safety and quality standards. It may be necessary to obtain a registration certificate issued by the Medical Devices Authority of India.
(3)Prepare documents and materials:Prepare documents and materials to support the export application, such as product manuals, quality assurance certificates, product test reports, production license certificates, registration certificates, product labels, business registration certificates,import and exportlicenses, etc.
(4)Consultation and agency:Consider cooperating with consulting companies or agencies with experience in India. They can provide professional guidance and handle complex procedures and paperwork.
Plant products exported to Russia should be accompanied by a certificate of origin issued by an authoritative institution to enjoy tariff preferences and reduce export costs.Fill out the application and submit documents:Fill out the export application and submit the required documents and materials according to the requirements of the relevant Indian agencies. Ensure that the information provided is accurate and submit the application on time.
(6)Review and approval:The relevant Indian agencies will review the application and the submitted documents, and may conduct inspections, tests, and evaluations to confirm that the product meets the standards and requirements. Once the review is passed, export approval will be obtained.
(7)Comply with the requirements of the importing country:In addition to Indias export requirements, it is also necessary to understand and comply with the import requirements of the target country, which may involve matters such as customs declaration, tariffs, taxes, import licenses, etc.
The above steps may vary depending on the product type, country, and regulatory changes. Therefore, before exporting, it is recommended to seek advice from professionals or communicate with local trade agencies to ensure that all necessary requirements are met.
Related Recommendations
Category case
Contact Us
Email: service@sh-zhongshen.com
Related Recommendations
Contact via WeChat

? 2025. All Rights Reserved. 滬ICP備2023007705號-2  PSB Record: Shanghai No.31011502009912
PSB Record: Shanghai No.31011502009912